Posted on February 24, 2021
A new Therapeutic Goods Administration (TGA) framework will commence on 25th February 2021, changing the definitions of personalised medical devices and the way they are regulated.
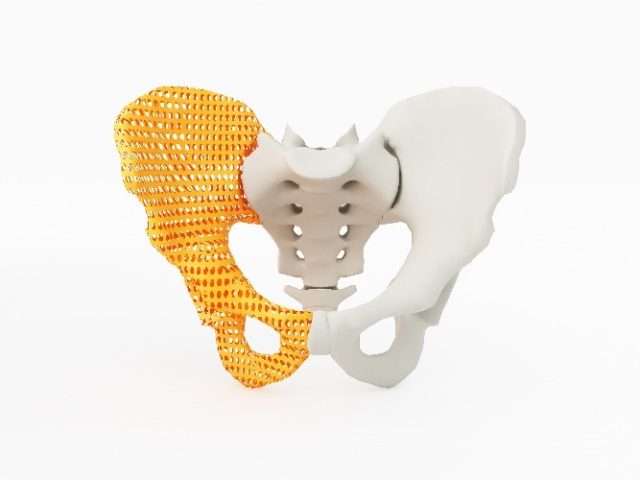
Personalised medical devices are medical devices produced to suit the needs of a particular individual. Previously, personalised medical devices included “Custom-made medical devices” and “Adaptable medical devices”, of which the former was exempt from inclusion in the Australian Register of Therapeutic Goods (ARTG).
The new regulatory framework continues to exempt custom-made devices from inclusion on the ARTG; the primary change being that the scope of products that meet the definition has been reduced by introducing new concepts; “Patient matched medical devices” and “Devices produced using the Medical Device Production System (MDPS)”. Both new types of devices will require inclusion on the ARTG from 25 February 2021. As a general guide, manufacturing, importing, or supplying more than five (5) of a kind of medical device in a year would make it unlikely for these devices to remain classified as “custom-made medical devices” under the new framework.
Patient matched medical devices are medical devices matched to a patient’s anatomy within a specified design envelope using techniques such as scaling of the device and are typically produced in batches through a controlled process. Manufacturers and suppliers with patient-matched medical devices will need to liaise with the TGA on transition arrangements by 25 August 2021 and inclusion applications will need to be submitted prior to 1 November 2024.
MDPS refers to a validated, multi-component design and production system that can be supplied to healthcare professionals and facilities for the production of specific patient-matched medical devices. Devices produced using MDPS will need to be included on the ARTG and supplied with instructions to enable the safe production of devices that meet their intended purpose.
In addition to the new definitions in the framework, while custom-made medical devices will continue to be exempt from inclusion on the ARTG, there are new obligations on the information to be supplied with the device, such as record keeping and reporting, and regulator inspection and review, which are required to be met from 25 February 2021.
If you are a manufacturer, supplier or sponsor affected by this change and you need assistance on the next steps, please contact our Regulatory, Quality & Compliance team at [email protected].